Chemical reactions between carbonates and pyrolite mantle and the origin of ultradeep diamonds
The presence of carbonates in inclusions in diamonds, coming from depths exceeding 670 km, is the obvious evidence that carbonates indeed exist in the Earth's lower mantle. However, their range of stability, crystal structures, the thermodynamic conditions of the decarbonation process in iron-bearing carbonates, and possible reactions between carbonates and mantle-forming silicates remain poorly constrained. One of the major goals of project 7 is to investigate the behaviour of iron-bearing carbonates at pressures up to 140 GPa and temperatures over 3000 K employing novel methods of high-pressure single-crystal X-ray diffraction and Mössbauer spectroscopy in laser-heated diamond anvil cells.
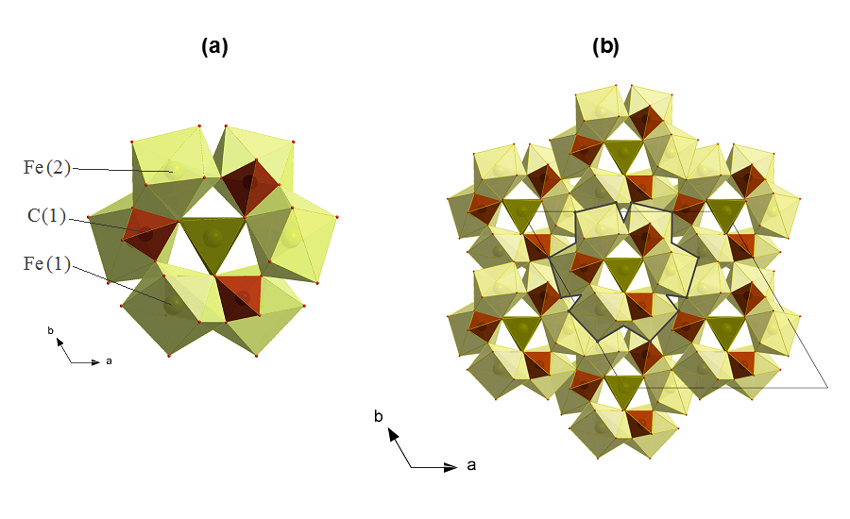
So far, the most important findings are related to the high-pressure synthesis and solving the structures of two new compounds – tetrairon (III) orthocarbonate, Fe43+C3O12, and diiron (II) diiron (III) tetracarbonate, Fe22+Fe23+C4O13. Both of these phases contain CO4 tetrahedra, and the former has a unique structure. This shows that high-pressure carbonates may not resemble other compounds (including silicates). Fe4C4O13 was found to be stable at conditions along the entire geotherm to the depth of at least 2500 km that demonstrates that self-oxidation-reduction reactions can preserve carbonates in the Earth's lower mantle.
Fig. 1 Crystal structures of high-pressure carbonates. (a) Tetrairon (III) orthocarbonate Fe4C3O12 and (b) diiron (II) diiron (III) tetracarbonate Fe4C4O13, at ambient temperature and 74(1) and 97(2) GPa, respectively. In (a), three FeO8 bicapped prisms (light green) and three CO4 tetrahedra (brown) form a ring with 3-fold symmetry by corner and edge sharing. The rings form layers that are stacked along the c-axis. FeO6 prisms (dark green) are connected by triangular bases and located in the channels created by the rings. In (b), FeO8 bi-capped prisms (blue) are connected in a 3-D framework by dimers of edge-shared FeO7 monocapped prisms (light green) and zigzag-shaped C4O1310- chains (brown).